Food Safety and Quality
GRI 3-3, 416-1, 417-1, SASB FB-MP-250a.2, FB-MP-250a.3, FP5, FP8
Food safety and quality are fundamental to our business and at the heart of everything we do. We are dedicated to continuously reviewing every aspect of our operations to make sure that we deliver safe, high-quality, and nutritious food products for our customers and consumers.

Our approach
Within our processing operations, we maintain a robust global food safety and quality program. This includes Standard Operating Procedures (SOPs), Sanitation Standard Operating Procedures (SSOPs), Hazard Analysis and Critical Control Points (HACCP) protocols, and validated technology interventions – each designed to eliminate or reduce biological, chemical, and physical hazards in raw material production. Many of our brands and business units also have specific policies dedicated to food safety and quality.
These processes are closely monitored by JBS team members trained in food safety and quality assurance. Government inspectors also review and verify our processes before approving products for distribution, assuring compliance with all applicable regulations.
All JBS business units implement the following strategies:
- Good Manufacturing Practices (GMP) program to support operational consistency and regulatory compliance.
- Recall and market withdrawal procedures that provide trace-back and trace-forward capabilities for accurate identification and management of affected products when necessary.
- A documented Pest Control Program and Food Security program.
Our Global Food Safety and Quality Assurance (FSQA) team is structured to provide strong oversight and consistent, industry-leading performance. Based in the U.S., our Global FSQA Department sets enterprise-wide guidelines, metrics, and priorities, which are deployed by local teams in each business unit. This streamlined reporting structure reflects the priority we place on food safety and quality, enabling swift, informed, and action-oriented decision-making.
BBest practices and KPIs are shared across the company during quarterly meetings hosted by the Global FSQA Department, as well as in more targeted routine meetings within specific brands and business units. These meetings provide a forum to exchange learnings, highlight current initiatives, and align on future priorities and targets.
Our business units also participate in external industry associations, councils, and working groups to advance consumer health and safety and accelerate progress. These include:
Food Safety Research
As part of our ongoing investment in research and innovation, we operate microbiological testing and biotechnology laboratories around the world. Equipped with state-of-the-art infrastructure, these labs allow us to track food safety metrics more precisely across our brands and accelerate testing timelines.
Our brands also partner with higher education institutions worldwide to invest in research and experiments aimed at improving consumer health and safety.
Trainings, Audits, and Performance Monitoring
GRI,416-1 SASB FB-MP-250a.2, FP5

Team Member Trainings
New JBS team members in relevant roles receive comprehensive training on quality assurance and food safety management systems during onboarding, with additional job-specific training provided as needed. All of our FSQA team members also receive specific HACCP and additional food safety and quality assurance training annually.
Third-Party Audits, Inspections, and Certifications
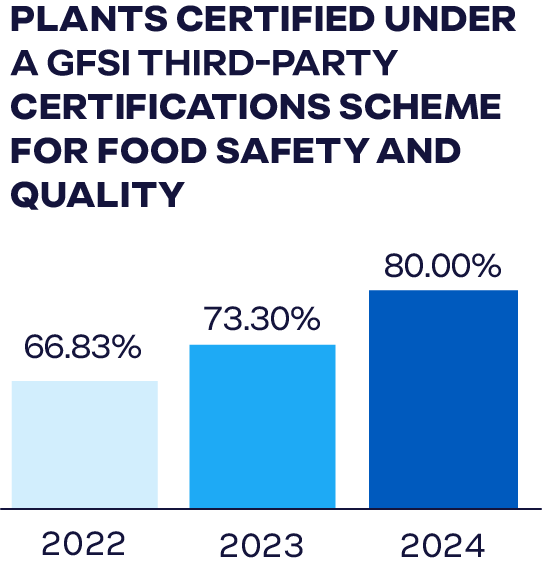
We conduct numerous food safety and quality system audits each year, using accredited, independent auditing firms. In 2024, 80% of our global facilities were audited and certified under schemes recognized by the Global Food Safety Initiative (GFSI), one of the most rigorous food safety benchmarks in the industry. This represents a 1% increase from the percentage of certified facilities in 2023.
In addition to these, our facilities undergo independent third-party audits based on the unique quality standards of our customers.


Internal Audits and Performance Monitoring
SASB FB-MP-250a.3
Our FSQA team members conduct internal audits and inspections to further evaluate food safety, quality assurance, and compliance across our operations. In 2024, we completed the fourth year of data collection and tracking against our internal Global Food Safety and Quality Assurance Scorecard. This tool helps benchmark performance indicators across global operations and business units, identify areas for improvement, and drive continuous progress.
The scorecard evaluates 11 metrics across four key pillars: regulatory compliance, animal welfare and prevention of antimicrobial resistance, customer and consumer satisfaction, and product safety and quality.
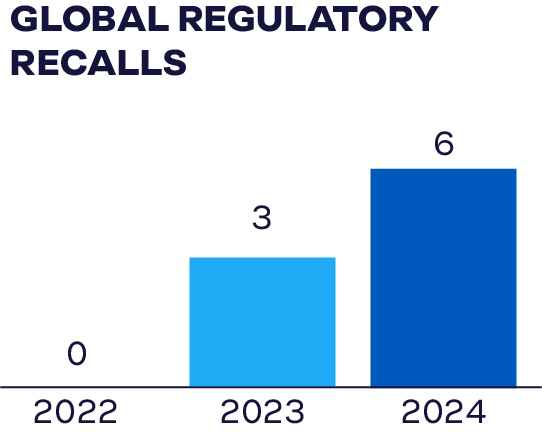
Although we have comprehensive systems in place to provide our customers and consumers with safe, high-quality food, in rare instances, a product recall may be necessary to protect public health. In these cases, we follow standardized procedures in collaboration with federal regulatory partners to quickly track and remove affected products from distribution channels. We communicate recall information to customers and consumers according to local government guidelines. In 2024, there were 6 regulatory recalls due to food safety, quality, or labeling in our global operations.
Case Studies
JBS Australia’s Pork business invested AUS$ 11.2 million in technology and equipment to enhance food safety and improve the quality of finished products. This investment included the installation of new carcass refrigeration chambers, auto bagging machines, auto stringing machines, inline de-rinders, and increased vacuum packaging capacity.
These advancements will result in less handling, extended shelf life, and reduced microbial growth, all contributing to the superior quality of JBS Australia’s finished pork products. In addition to food safety and quality improvements, these investments are also expected to reduce plastic waste, increase yields, improve process efficiencies, and generate greater cost savings.

